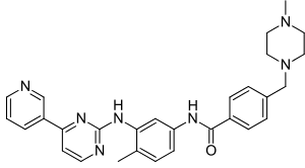
Imitinef Mercilet or Imatinib is a drug used treat certain types of cancer. Imatinib works by overcrowding an abnormal enzyme feature of the disease. Imatinib is the first accepted drug to directly turn off the signal of a protein well-known to cause a cancer. Imatinib is swiftly engrossed when given by mouth, as well as is highly bioavailable, 98% of an oral dose reach the bloodstream. Metabolism of imatinib occur in the liver .Though the long-term side effects of imatinib contain not yet been ascertain, research suggests that it is usually very in good health tolerated.
The most common side effects include:
Imatinib is rapidly absorbed when given by mouth, and is highly bioavailable and 98% of an oral dose reaches the bloodstream. Metabolism of imatinib occurs in the liverand is mediated by several isozymes of the cytochrome P450 system, including CYP3A4 and to a lesser extent, CYP1A2, CYP2D6, CYP2C9, and CYP2C19. The main metabolite, N-demethylated piperazine derivative, is also active. The major route of elimination is in the bile , feces and only a small portion of the drug is excreted in the urine. Most of imatinib is eliminated as metabolites. only 25% is eliminated unchanged. The half-lives of imatinib and its main metabolite are 18 and 40 hours, respectively. It blocks the activity of Abelson cytoplasmic tyrosine kinase (ABL), c-Kit and the platelet-derived growth factor receptor (PDGFR). As an inhibitor of PDGFR, imatinib mesylate appears to have utility in the treatment of a variety of dermatological diseases. Imatinib has been reported to be an effective treatment for FIP1L1-PDGFR alpha mast cell disease, hypereosinophilic syndrome, and dermatofibrosarcoma protuberans
- feeling sick (nausea), diarrhea, headaches, leg aches/cramps, fluid retention, visual disturbances, itchy rash, lowered resistance to infection, bruising or bleeding, loss of appetite, weight gain, reduced number of blood cells (neutropenia, thrombocytopenia, anemia), and edema.
- Severe congestive cardiac failure is an uncommon but recognized side effect of imatinib and mice treated with large doses of imatinib show toxic damage to their myocardium.
- If imatinib is used in prepubescent children, it can delay normal growth, although a proportion will experience catch-up growth during puberty.
Imatinib is rapidly absorbed when given by mouth, and is highly bioavailable and 98% of an oral dose reaches the bloodstream. Metabolism of imatinib occurs in the liverand is mediated by several isozymes of the cytochrome P450 system, including CYP3A4 and to a lesser extent, CYP1A2, CYP2D6, CYP2C9, and CYP2C19. The main metabolite, N-demethylated piperazine derivative, is also active. The major route of elimination is in the bile , feces and only a small portion of the drug is excreted in the urine. Most of imatinib is eliminated as metabolites. only 25% is eliminated unchanged. The half-lives of imatinib and its main metabolite are 18 and 40 hours, respectively. It blocks the activity of Abelson cytoplasmic tyrosine kinase (ABL), c-Kit and the platelet-derived growth factor receptor (PDGFR). As an inhibitor of PDGFR, imatinib mesylate appears to have utility in the treatment of a variety of dermatological diseases. Imatinib has been reported to be an effective treatment for FIP1L1-PDGFR alpha mast cell disease, hypereosinophilic syndrome, and dermatofibrosarcoma protuberans
 RSS Feed
RSS Feed

